Sodium Carbonate (Washing Soda), Na2CO3⋅10H2O Sodium carbonate
By A Mystery Man Writer
Description
Sodium Carbonate (Washing Soda), Na2CO3⋅10H2O
Sodium carbonate is generally prepared by Solvay Process. In this process, advantage is taken of the low solubility of sodium hydrogencarbonate whereby it gets precipitated in the reaction of sodium chloride with ammonium hydrogencarbonate. The latter is prepared by passing CO2 to a concentrated solution of sodium chloride saturated with ammonia, where ammonium carbonate followed by ammonium hydrogencarbonate are formed. The equations for the complete process may be written as :
2NH3+H2O+CO2→(NH4)2CO3(NH4)2CO3+H2O+CO2→2NH4HCO3NH4HCO3+NaCl→NH4Cl+NaHCO3
Sodium hydrogencarbonate crystal separates. These are heated to give sodium carbonate.
Video solution 1: Sodium Carbonate (Washing Soda), Na2CO3⋅10H2O Sodium carbonate is generally prepared by Solvay Process. In this process, advantage is taken of the low solubility of sodium hydrogencarbonate whereby it gets precipitated in the reaction of sodium chloride with ammonium hydrogencarbonate. The latter is prepared by passing CO2 to a concentrated solution of sodium chloride saturated with ammonia, where ammonium carbonate followed by ammonium hydrogencarbonate are formed. The equations for the complete process may be written as : 2NH3+H2O+CO2→(NH4)2CO3(NH4)2CO3+H2O+CO2→2NH4HCO3NH4HCO3+NaCl→NH4Cl+NaHCO3 Sodium hydrogencarbonate crystal separates. These are heated to give sodium carbonate.
Video solution 1: Sodium Carbonate (Washing Soda), Na2CO3⋅10H2O Sodium carbonate is generally prepared by Solvay Process. In this process, advantage is taken of the low solubility of sodium hydrogencarbonate whereby it gets precipitated in the reaction of sodium chloride with ammonium hydrogencarbonate. The latter is prepared by passing CO2 to a concentrated solution of sodium chloride saturated with ammonia, where ammonium carbonate followed by ammonium hydrogencarbonate are formed. The equations for the complete process may be written as : 2NH3+H2O+CO2→(NH4)2CO3(NH4)2CO3+H2O+CO2→2NH4HCO3NH4HCO3+NaCl→NH4Cl+NaHCO3 Sodium hydrogencarbonate crystal separates. These are heated to give sodium carbonate.

Washing soda has the formula Na_(2) CO_(2).10H_(2)O. What is mass of anhydrous sodium carbonate

What Is Sodium Carbonate?

Washing soda has the formula Na_(2) CO_(2).10H_(2)O. What is mass of anhydrous sodium carbonate

What is Sodium Carbonate / Washing Soda / Soda Ash Chemical Formula
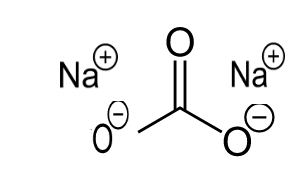
the story of washing soda na2co3 10h2o - Overview, Structure, Properties & Uses

Washing Soda Formula- Chemical Name is na2co3

10-hydrate sodium carbonate na2co3.10h2o ar grade
Wintersun code: 19-011-003 CAS Number: 497-19-8 Molecular Formula: Na2CO3 Sodium Carbonate (Na2CO3): 99.7 % min Sodium Oxide (Na2O):58.3 % min Sodium

Sodium Carbonate Dense Soda Ash [Na2CO3] [CAS_497-19-8] +99.7%, White Crystals (55.12 Lb Bag)

Sodium Carbonate (Na₂CO₃) - Synthesis of Washing Soda, Applications and Uses - Infinity Learn by Sri Chaitanya

Washing Soda Property, Benefits and Preparation of Sodium Carbonate

How to Balance: Na2CO3 + HCl = NaCl + H2O + CO2
Appearance: White Solid Sodium Carbonate uses: An intermediate for the production or for the treatment of a wide variety of products in the chemicals

Sodium Carbonate (Soda Ash Dense) [Na2CO3] [CAS_497-19-8] 99.6+%, White Solid (50 Lb Bag) by Wintersun Chemical
from
per adult (price varies by group size)







