14.6: Buffers - Chemistry LibreTexts
By A Mystery Man Writer
Description
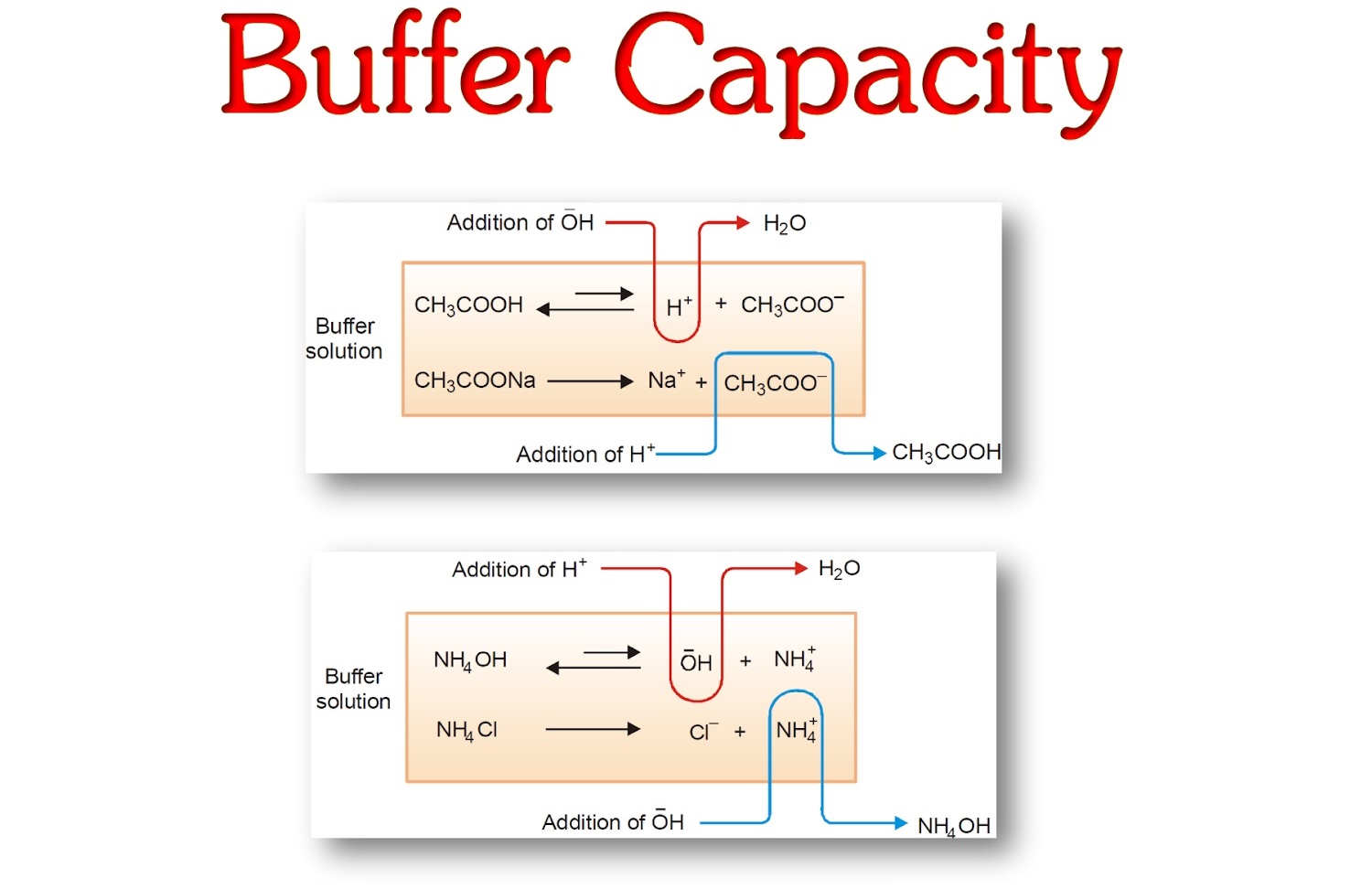
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the …
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the hydronium ion concentration of a buffer solution does not change greatly when a small amount of acid or base is added to the buffer solution. The base (or acid) in the buffer reacts with the added acid (or base).
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the hydronium ion concentration of a buffer solution does not change greatly when a small amount of acid or base is added to the buffer solution. The base (or acid) in the buffer reacts with the added acid (or base).
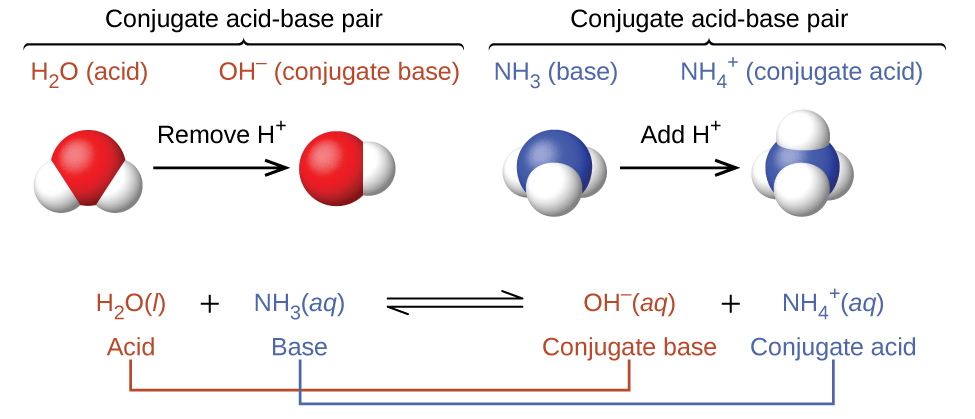
14.1: Brønsted-Lowry Acids and Bases - Chemistry LibreTexts

Buffers - Chemistry LibreTexts, PDF, Buffer Solution
8.2: Formation Constants Lab - Chemistry LibreTexts
Introduction to Chemistry - Chemistry LibreTexts
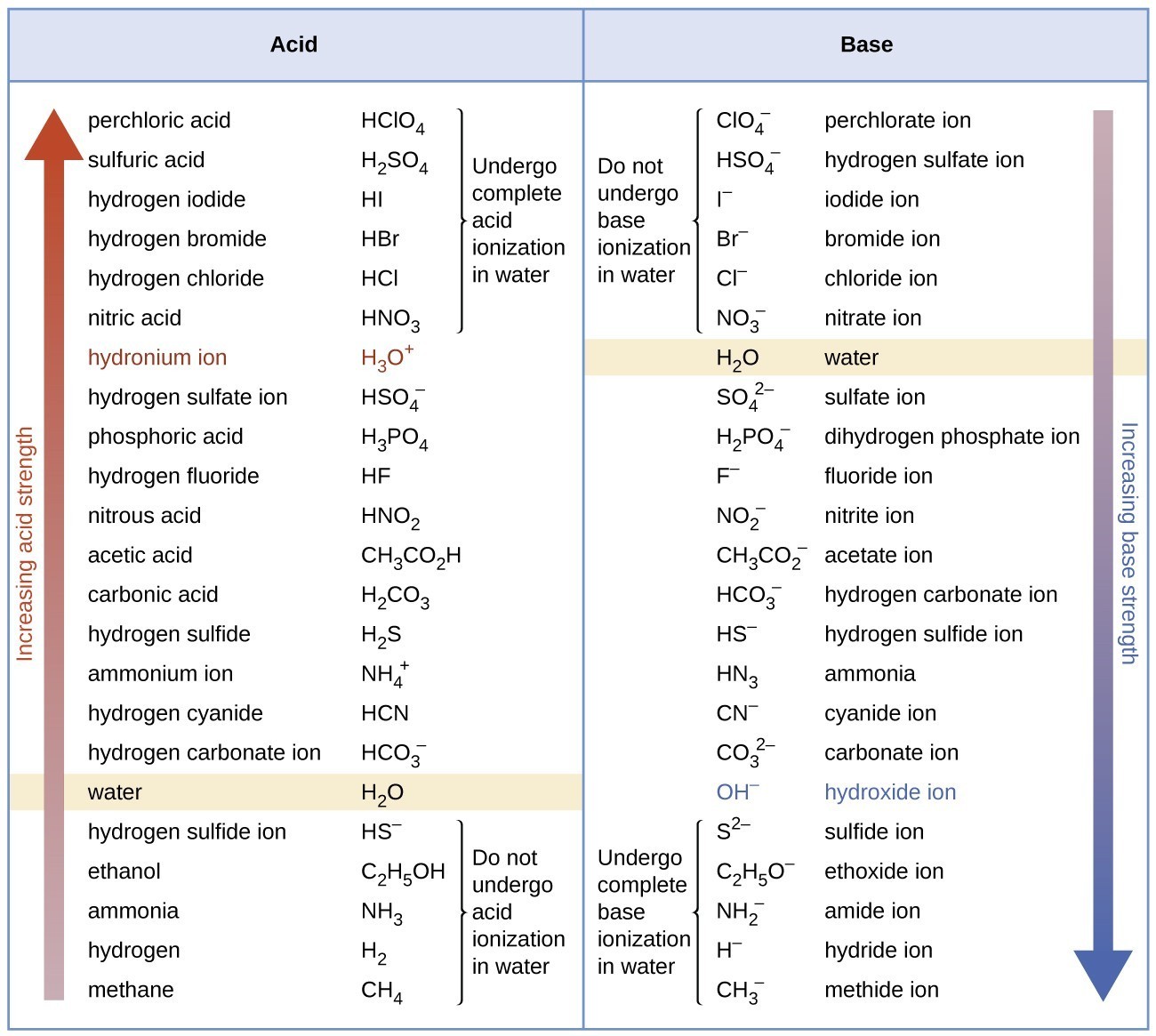
14.3: Relative Strengths of Acids and Bases - Chemistry LibreTexts
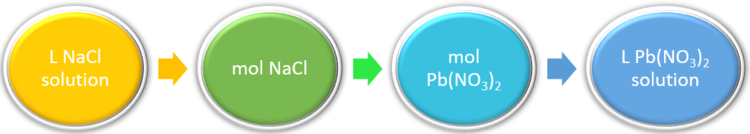
13.8: Solution Stoichiometry - Chemistry LibreTexts
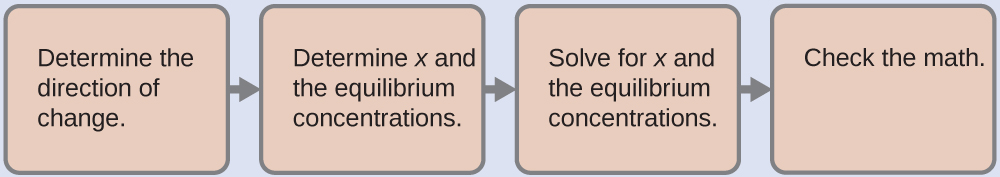
14.6: Buffers - Chemistry LibreTexts
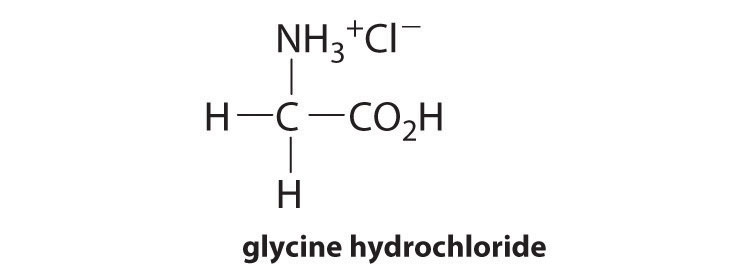
4.E: Buffer, Solubility, Common Ion Effects, and Acid-Base
4.E: Buffer, Solubility, Common Ion Effects, and Acid-Base
Introduction to Chemistry - Chemistry LibreTexts

Why do we have to assume that the base is weak if we add a strong
10.5: Buffers - Chemistry LibreTexts
from
per adult (price varies by group size)